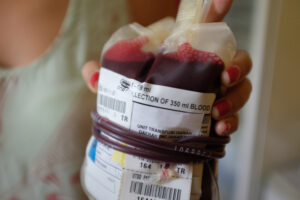
Observational studies suggest that prehospital whole blood may offer benefits over blood components in trauma patients, including a reduction in the total volume of blood products transfused. The SWiFT (Study of Whole Blood in Frontline Trauma) randomized controlled superiority trial included 616 trauma patients with life-threatening bleeding (75% male; 71% blunt injuries) in England, who were randomized to receive either two units of whole blood (n=314) or standard care (two units of red cells and two units of plasma) (n=302) in air ambulances. The percentage of patients who died or received a massive transfusion (>10 units of blood) within 24 hours of randomization was similar—48.7% in the whole blood arm and 47.7% in the standard care arm (relative risk, 1.02; 95% C.I., 0.30 to 1.31; p=0.84). Prothrombin times, however, exceeded the normal range in 41% of the patients who received whole blood compared to 31% who received standard care. There was no impact on clinical outcomes. No differences in mortality were observed 30 or 90 days after randomization between arms, and safety profiles were similar. As no substantial differences were observed between prehospital whole blood and blood components in trauma patients, other factors—such as logistical advantages, cost, and overall blood availability—should be considered when selecting
Reference: